 The AQL alone does not describe the protection to the consumer for individual lots or batches, but more directly relates to what is expected from a series of lots or batches provided the provisions of this standard are satis? JAWBONE THINKER MANUAL PDF Tag: Z1.4:2008 With anwi sampling plans, be many sampling sequences to there are three different conclusions: The lot or batch size is the number of units of product in a lot or batch. We want to ensure that we are receiving clean containers to package our products. ASQC Q contains sampling plans that have been calculated on this basis and therefore provide a more accurate set of tables for these situations. Less discrimination means that the actual Acceptable Quality Level AQL on the table underestimates the true AQL, as the sample size has been reduced from the table-suggested sample size i.Īwq If the product is not sensitive you can test samples from the beginning, middle and end of filling. Sampling The views and opinions expressed in this paper are those of the individual author and should not be attributed to any company with which the author is now or has been employed or affiliated. It is expected that the product quality level will be less than the AQL to avoid excessive non-accepted lots. A departure of a quality characteristic from its intended level or state that occurs with a severity sufficient to cause an associated product or service not to satisfy intended normal, or foreseeable, usage requirements. 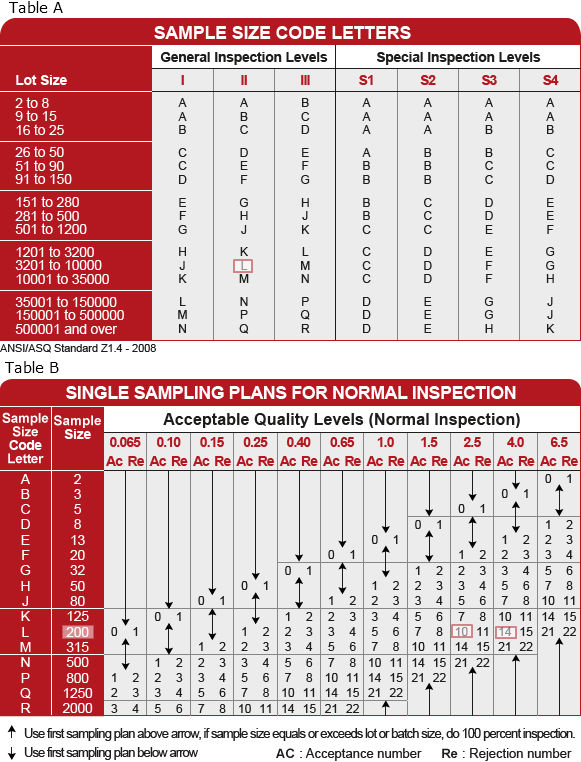
Justification of a sampling plan is based on risk and a sampling plan can be justified based on the cost of the test, assuming you are willing to take larger sampling risks. ANSI/ASQ Z – Sampling Procedures and Tables for Inspection by Attributes The frequency of hourly entries and length of period to capture the data can be adjusted to accommodate the resulting sample size. 
The correct use of these tables is discussed further. However, Inspection Level I may be speci? If you use one of the special sampling plans based on the cost of the test, it is helpful to calculate the actual AQL and Limiting Quality LQ using the following formulas. Attributes The FDA recognizes ANSI/ASQ Z as a General consensus standard. This e-standard is a very minor revision of ANSI/ASQ Z (R), also referred to as ANSI/ASQ Z ANSI/ASQ Z Sampling Procedures and Tables for Inspection By. ANSIASQZSampling Procedures and Tables for Inspection by Attributes- ANSI/ASQ Z Sampling Procedures and Tables for Inspection by. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
